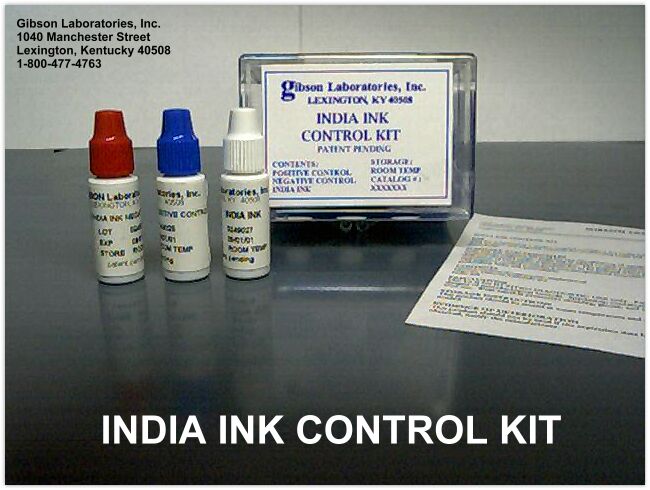
INDIA INK QC CONTROL KITS
INDIA INK WET MOUNT CONTROLS
INDIA INK POSITIVE CONTROL - CATALOG 100111 $50.00
EACH POSITIVE CONTROL CONTAINS SUSPENSIONS OF ENCAPSULATED CRYPTOCOCCUS neoformans AS A POSITIVE CONTROL
INDIA INK NEGATIVE CONTROL - CATALOG 100112 $50.00
EACH NEGATIVE CONTROL CONTAINS A SUSPENSION OF NON- ENCAPSULATED YEAST
INDIA INK 1ML - CATALOG 100114 $50.00
CONTAINS 1ml OF INDIA INK FOR WET MOUNT PREPARATIONS
INDIA INK KIT - CATALOG 100115 $140.00
KIT CONTAINS 1 EACH OF NEGATIVE CONTROL, POSITIVE CONTROL AND THE INDIA INK
India Ink Control Product Information
Recommended to be part of Quality Control to monitor India Ink Wet Mount Preparations.
Patent Pending
India Ink Wet Mount ControlsPositive Control: Each positive control contains 1ml suspension of encapsulated Cryptococcus neoformans.
Negative Control: Each negative control contains 1ml suspension of non-encapsulated yeast.
India Ink: Contains 1 ml of India Ink for use with wet mount preparations.
SIMPLE PROCEDURE
1. Mix equal parts of Controls with India Ink on slides.
2. Add cover slips.
3. Examine at X100 and X1,000.
Also available: Demonstration Suspensions and Supplies.
INDIA INK CONTROL
USE
India Ink Control is recommended to be part of quality control to monitor India Ink wet mount preparations.
SUMMARY AND EXPLANATION
India Ink wet mounts are used for the visualization of encapsulated microorganisms, especially Cryptococcus neoformans, directly in clinical specimens. The polysaccharide capsules of organisms will exclude the particles of ink, and the capsules will appear as clear haloes around the organisms on semi-opaque backgrounds.
PRINCIPLES
The India Ink Positive Control consists of a suspension of encapsulated Cryptococcus neoformans as a positive control for India Ink wet mount preparations. A suspension of non-encapsulated yeasts is provided as a negative control for India Ink wet mount preparations.
COMPONENT
Positive: Cryptococcus neoformans (encapsulated suspension)
Negative: Yeasts (non-encapsulated suspension)
PRECAUTIONS
This medium is for IN VITRO DIAGNOSTIC USE only. Precautions should be taken against the dangers of microbiological hazards. Specimens, containers, and media should be sterilized after use. This product may contain potentially viable organisms. Handle it as you would a known pathogen.
STORAGE INSTRUCTIONS
Media should be stored at room temperature in the dark and remain in the original packaging until needed.
EVIDENCE OF DETERIORATION
This medium should not be used if the expiration date has passed. If any deficiencies are observed, notify the manufacturer.
OTHER MATERIAL REQUIRED BUT NOT SUPPLIED
The usual clinical microbiological equipment such as India Ink, Nigrosin, slides, cover slips, and microscope, are needed for procedures involving the use of this product.
PROCEDURE
1. Place equal parts of the India Ink Control suspension and India Ink (or Nigrosin) on the slide.
2. Mix well, and add a cover slip.
3. Examine at x100 to x1,000.
EXPECTED RESULTS
The India Ink Positive Control contains capsules that will exclude the ink and appear as clear haloes around the organism.
LIMITATION OF THE PROCEDURES
Results may be hindered if the emulsion with the suspension is too thick, blocking the transmission of light completely.
REFERENCES
1. Murray, P.R. et al. 1995. Manual of Clinical Microbiology, 6th ed. ASM. Washington, DC.
Gibson Laboratories, Inc.
1040 Manchester Street • Lexington, KY 40508
| © Lakewood Biochemical Co., Inc. Dallas, TX 75214 |
0415
Index , Alphabetical by Subject Titles
GENERAL CATALOG SECTIONS